Entropy definition biology8/31/2023  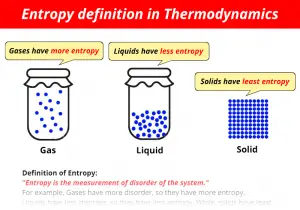
Large granules of glycogen are especially abundant in the liver and muscle cells. Glycogen - polysaccharide composed exclusively of glucose units used to store energy in animal cells. Takes into account changes in both energy and. So in order to maintain this highly organized state we need to be constantly spending energy to maintain the highly organized cells of our bodies.įree energy(G) - energy that can be extracted from a system to drive reactions. One of the laws of the universe is that the universe hates order and it's called this love of disorder. Where ΔH is the enthalpy of base stacking interactions adjusted for helix initiation factors ΔS is the of base stacking adjusted for helix initiation factors and for the contributions of salts to the R is the universal gas constant. īase-stacking calculation : Most accurate, but complicated Membrane proteins are not involved in passive diffusion.Īpart from concentration gradient, kinetic energy or natural of molecules further fuels the process.Įxample of simple diffusion: passive transport of small nonpolar molecules across the plasma membraneĮxample of facilitated diffusion: passive transport of glucose and ions into and out of the cell. The transport process is influenced by the characteristics of the transport substance and the nature of the bilayer. The energy from the sun is being converted to chemical energy, thereby producing organic compound (like glucose).Īs mentioned above, passive diffusion is a spontaneous phenomenon that increases the of a system and decreases the free energy. Basically, sun is the primary source of energy. In chemical terms, this means that they seek to have a stable number of electrons in their outermost orbital.Īll organisms require constant supply of energy in order to keep bodily functions and to fight against. To increase stability, atoms tend to react so that they lower their energy and increase their (randomness or lack of organization). The confusion was originated when some properties associated with the were subordinated like alternatives to explain biotic features for example, order, complexity, etc. Moddemeijer R: On Estimation of and Mutual Information of Continuous Distributions. H can be thought of as a measure of the average information (in bits) available per position that distinguishes an alignment from chance. H is the relative of the target and background residue frequencies. A measure of the degree of disorder within a system, which is related to the energy of a system unavailable for mechanical work. How to Prepare a Sodium Hydroxide SolutionĮqual to the times the ab solute temperatureĭepartment of Biochemistry and Molecular Biophysics The guy who wrote this is a chemistry professor who has tons of experience teaching thermodynamics to non-scientists. Thorough explanation of the first and second laws of thermodynamics. As energy is transferred from one form to another, some is lost as heat as the energy decreases, the disorder in the system&emdash and thus the &emdash increases.Įnzymes Protein molecules that act as catalysts in biochemical reactions. For example, in the case of oxygen binding to hemoglobin, the first oxygen has four different available binding sites.Ī measure of the disordered, degraded energy that is unavailable for work.Įnzyme A Protein produced by living organisms that acts as a catalyst for a specific biochemical (metabolic) reaction. 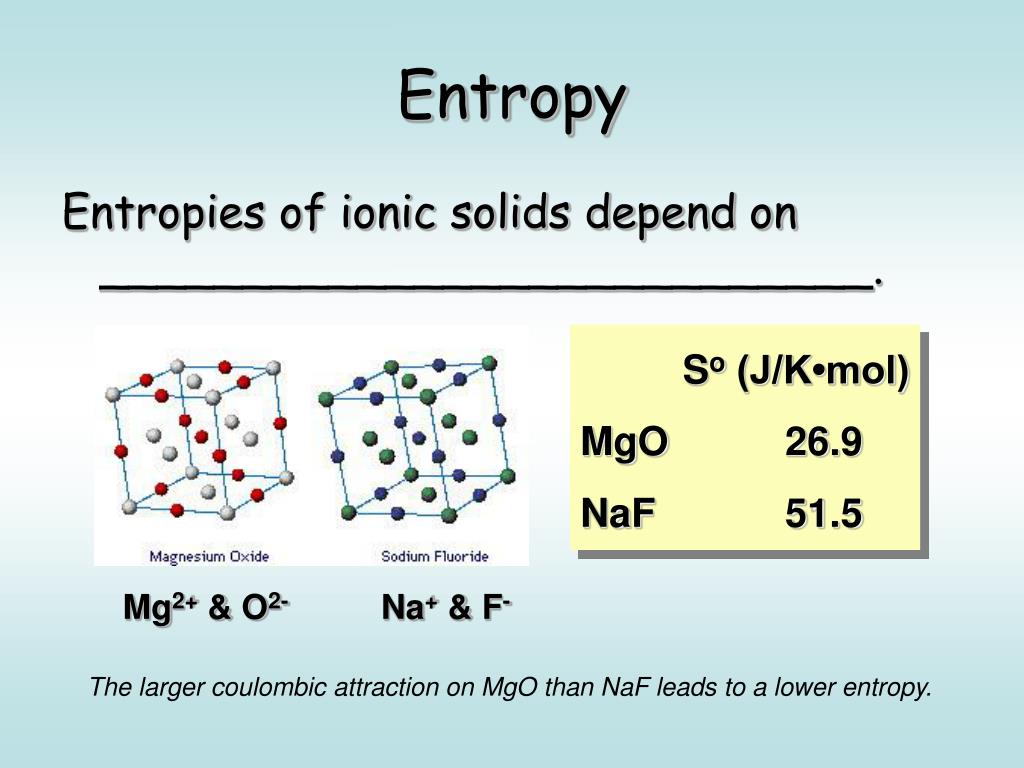
In all of the above types of cooperativity, entropy plays a role. 
The acceleration of a re action that occurs when reactive groups are constrained in a productive orientation, either intermolecularly as on an enzyme surface, or intramolecularly as in a model compound. A measure of the degree of dis order or randomness in a system the higher the entropy, the greater the disorder. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
